Projects
IMAGINE 2018
IMAGINE is an international study of postoperative ileus and provision of management after surgery. It is being coordinated by EuroSurg with STORCC acting as the local lead on the Central Coast. All STORCC Collaborators achieved a PUBMED citation from the research publications
IMAGINE seeks to profile gastrointestinal recovery after colorectal surgery. The secondary outcome is to assess the role of NSAIDs in expediting GI recovery and their respective safety.
Resources
STORCC Team Members for IMAGINE Study:
Local Lead: A Dawson
Consultants: A Dawson (Gosford Hospital), K Kwok (Gosford Private Hospital), P Stewart (Wyong Hospital)
Collaborators:
N Taylor, C Stevenson, A Drane, U Pahalawatta, LT Lai, A Debiasio (Gosford Hospital) A Nair, V Thirugnanasundralingam, K Muir (Wyong Hospital) J Wong, HJS Jun, R Hengpoonthana (Gosford Private Hospital)
The study ran with great success involving 4164 patients in the final dataset. The key findings from this study were:
The use of postoperative NSAIDs did not reduce the time to GI recovery.
The use of postoperative NSAIDs, when used between post-operative days 1-3 appeared safe.
Publications:
Eurosurg Collaborative. Safety and efficacy of non‐steroidal anti‐inflammatory drugs to reduce ileus after colorectal surgery. BJS. 2020; 107 (2): e161-e169. PMID: 31595986. DOI: http://dx.doi.org/10.1002/bjs.11326. (Main Paper)
Eurosurg Collaborative. Safety of hospital discharge before return of bowel function after elective colorectal surgery. BJS. 2020; 107 (5): p552-p559. PMID: 31976560. DOI: http://dx.doi.org/10.1002/bjs.11422. (Satellite Paper)
Eurosurg Collaborative. Timing of nasogastric tube insertion and risk of postoperative pneumonia: international, prospective cohort study. Colorectal Dis. 2020; [epub ahead of print]. DOI: http://dx.doi.org/10.1111/codi.15311. (Satellite Paper)
Eurosurg Collaborative: Validation of the OAKS prognostic model for acute kidney injury after gastrointestinal surgery', BJS OPEN 2022 https://doi.org/10.1093/bjsopen/zrab150
Worldwide locations of the contributing sites for the IMAGINE trial
BEACCHES
Many medical schools around the world run wilderness first responder training as part of experiential education. In 2018, the Central Coast Medical School (CCMS) launched BEACCHES (Beginning Education at Central Coast Hospitals) to provide first responder training that is specific to the Central Coast. This has been developed by CCMS in partnership with local surf life-saving organisations.
BEACCHES is a two-day intensive program where students will learn about critical medical and evacuation decisions and medical responses relevant to coastal or remote locations. Training and demonstrations will involve realistic scenarios with mock patients to challenge the students to integrate their learning. BEACCHES also provides opportunities for students to connect with their peers, support team, supervisors and the Central Coast locality.
Although BEACCHES ran in 2020, it was on hold during 2021 due to the COVID pandemic.
Publications:
Short B, Lambeth L, David M, Ryall M, Hood C, Pahalawatta U, Dawson A, 'An immersive orientation program to improve medical student integration and wellbeing', The Clinical Teacher, 16 323-328 (2019) DOI10.1111/tct.13077
STITCCH
Surgical Trainee Initiated Teaching in Central Coast Hospitals
The STITCCH programme is a teaching initiative aimed at attracting junior doctors to the education of senior medical students. It seeks to identified incentives and barriers to teaching at a JMO level, and provided curriculum packs, mentorship and logistical support to help overcome these barriers.
STITCCH commenced in 2015 and developed a suite of curriculum packs available for its tutors and is also reproduced free of charge for use worldwide on its website.
STITCCH has presented the results of its effectiveness at multiple national and international conferences, and received an award for Excellence in Clinical Teaching by the University of Newcastle.
STITCCH promoted a culture of near peer teaching at the Central Coast Local Health District with the development of multiple similar programs:
Promoting Residents Active Involvement in Student Education. PRAISE was commenced in 2018 following the success of STITCCH. This initiative aims to provide practical teaching for junior doctors in how to teach.
Resident Initiated Surgical Skills. RISSK was designed to improve procedural skills among medical students whilst improving procedural skill teaching capacity is now part of the regular NPMT curriculum.
Resident Led Surgical Exam Teaching. RESET commenced in 2019 and is a resident led program to provided near peer teaching for junior doctors sitting the GSSE exam for RACS. This has expanded in 2020 to cover Royal North Shore Hospital.
Due to its popularity, STITCCH has now rebranded to cover all specialities as the Near Peer Medical Teachers (NPMT (npmteaching.com)
Publications:
Panozzo B, Dixson G, St Flour P, Ryall M-A, Dawson A, 'Evaluating medical student and teacher attitudes of virtually delivered near peer medical teaching during the COVID-19 pandemic', Australian Journal of Clinical Education, 2021 DOI10.53300/001c.27483
Respiratory COmplications after abdomiNal surgery (RECON) trial 2019
STARSurg Collaborative
STARSurg is an international collaborative network of students and surgical trainees, supported by consultant surgeons and anaesthetists. The group aims to engage collaborators with common research goals to deliver meaningful studies across multiple countries and the UK and Ireland.
Background
Post-operative pulmonary complications (PPCs) are a common complication of major abdominal surgery and associated with increased morbidity, mortality and length of hospital stay. Royal College of Anaesthetists (RCOA) and Enhanced Recovery After Surgery (ERAS) guidelines provide pre-, intra- and post-operative measures to reduce risk of PPCs.
Study aims
Primary: Explore the incidence of PPCs following major abdominal and incisional hernia surgery
Secondary: (1) Evaluate adherence to peri-operative measure to reduce risk of PPCs; (2) establish risk factors for development of PPCs
Outcome measures
Primary: Adherence to perioperative guidelines (RCOA, ERAS) for the prevention of PPCs
Key secondary: Incidence of PPCs within 7 days of surgery, defined using StEP definitions
Data collection
Data collected using an online web application (REDCap), widely used and secure data capture service.
Data analysis
Data will be pooled and analysed centrally and anonymously with no surgeon- or hospital- level analysis.
Mini-team structure
Mini-teams of 1 to 2 medical students and one junior doctor per speciality group per data collection period, at each participating centre will prospectively collect data. All mini-teams should be supervised by a consultant in surgery and/or anaesthesia or critical care. A hospital lead may be appointed in each hospital to hold overall responsibility for site registration and coordinating handover between teams.
Audit periods
Four consecutive 2-week periods from 21st Jan 2019 - 17th Mar 2019 (with additional 30-day follow-up)
Inclusion criteria
Consecutive adult patients undergoing major abdominal surgery across gastrointestinal, hepatopancreatobilary, urology, vascular, gynaecology and transplant surgery through an abdominal incision, including one or more of: (i) Visceral resection; (ii) Reversal of stoma; (iii) Open repair of abdominal aortic aneurysm or vascular bypass procedure; (iv) Anterior abdominal wall incisional hernia or parastomal hernia repair; (v) Transplant surgery. A full list of included procedures in available in the RECON Study Protocol. Any operative approach (open, laparoscopic, robotic) and indication is eligible.
Exclusion criteria
Abdominal surgery without resection; Caesarean sections; Groin and primary (not incisional) abdominal wall hernia repair; Return to theatre (each patient only eligible for inclusion once); Vascular surgery with a groin or limb incision only.
The study was immensely successful with 11,251 patients included in the final dataset, the largest for a STARSurg project to date. Key findings of the current analyses are as follows:
The national adjusted PPC rate was 7.5%
Substantial variation was seen across all specialties ranging from 3.0% (n = 60/1983) in gynaecological to 17.4% (n = 136/668) in Upper GI procedures.
There is further analysis of the RECON dataset occurring with aims to explore:
Associations between interventions and PPC rates.
Variations in specialty specific PPC rates.
Measures to be introduced as a cost-effective effort to reduce PPCs.
STARSurg will shortly submit a manuscript based on the RECON dataset. (RECON (2018/19) – STARSurg UK)
Collaborators:
Bourke E, Chong A, Clayton S, Dawson A, Hardy E, Iqbal R, Le L, Mao S, Marinelli I, Metcalfe H, Panicker D, R HH, Ridgway S, Tan HH, Thong S, Van M, Woon S*, Woon-Shoo-Tong XS, Yu S (Gosford Hospital); Chinta SH, Dawson A, Li J, Phan J, Rahman F, Segaran A, Shannon J, Woon-Shoo-Tong XS*, Zhang M (Wyong Hospital).
Publications:
Evaluation of prognostic risk models for postoperative pulmonary complications in adult patients undergoing major abdominal surgery: a systematic review and international external validation cohort study', The Lancet Digital Health, 4 e520-e531 (2022) https://doi.org/10.1016/S2589-7500(22)00069-3
Death following pulmonary complications of surgery before and during the SARS-CoV-2 pandemic British Journal of Surgery Volume 108, Issue 12, December 2021, Pages 1448–1464, https://doi.org/10.1093/bjs/znab336 13 Nov 2021
Preliminary model assessing the cost-effectiveness of preoperative chlorhexidine mouthwash at reducing postoperative pneumonia among abdominal surgery patients in South Africa PLOS ONE 2021 https://doi.org/10.1371/journal.pone.0254698
GlobalSurg 3: Quality and Outcomes after global cancer surgery: a prospective, international cohort study. 2019
GlobalSurg Collaborative
Background: Of the 15.2 million individuals diagnosed with cancer in 2015, over 80% will need surgery (1). In tumours amenable to surgical resection, surgery often offers the best chance of cure, particularly in early-stage disease. It has been estimated that 45 million surgical procedures are needed each year worldwide, yet, fewer than 25% of patients with cancer have access to safe, affordable, and timely surgery. While death rates from cancer are decreasing in high-income countries, the opposite has been demonstrated in low- and middle-income countries (LMICs) (2). Up to 1.5% of the gross domestic product is lost because of cancer in some LMIC regions (3).
General surgeons manage patients with the most common cancers on a day-to-day basis. Breast cancer (global incidence ranked 1st, global mortality ranked 5th), gastric cancer (incidence ranked 5th, mortality ranked 3rd), and colorectal cancer (incidence ranked 3rd, mortality ranked 2nd), represent a significant burden of disease across income settings (1). Yet, most studies that examine the global distribution and outcomes of solid cancers use simulated methods due to the absence of robust data, including country-specific epidemiological data, stage distribution, and treatment approaches (1).
Aim: The aim is to determine the variation in quality of cancer surgery worldwide. Quality will be determined using measures covering infrastructure, care processes, and outcomes. We will concentrate on the most common surgically-treated cancers worldwide: breast, gastric and colorectal cancer. The primary aim focusses on 30-day mortality and complication rates after cancer surgery. The secondary aim is to characterise infrastructure and care processes in the treatment of these cancers worldwide.
Primary outcome measure: 30-day mortality and complication rates after cancer surgery.
Primary comparison: Between country groups defined by human development index.
Study Participants: Medical students, doctors, nurses and research officers will form data collection teams. Any hospital, anywhere in the world performing emergency or elective surgery for breast, gastric or colon cancer is eligible to join Study Periods: Any 4- week data period (plus 30-day follow-up) between 1st April and 31st October 2018
Authorship: All collaborators will receive PubMed citable authorship according to the following model https://doi.org/10.1016/S1473-3099(18)30101-4
For more information about GlobalSurg please see http://globalsurg.org/who-we-are/
Read the full protocol: http://globalsurg.org/gs3
STORCC Team Members for GlobalSurg 3 Study:
Local Leads: A Dawson, Xiao-Ming Woon Shoo Tong, Andrew Drane
Consultants: A Dawson (Gosford Hospital), R Poon (Gosford Private Hospital), P Stewart (Wyong Hospital)
Collaborators: U Pahalawatta, Bryan Lim, Jacqueline Phan, Xiao-Ming Woon-Shoo-Tong, Andea Yeoh (Gosford Hospital) Angela Au, Ashe DeBiasio, Idy Deng, Jananee Myooran, Amrita Nair (Wyong Hospital) Lilian Charman, Andrew Drane, Sharon Laura, Charmaine Chu Wen Lo, Amy Mozes, Hao Han Tan, Ellen Wall (Gosford Private Hospital)
Data Validators: Log Tung Lai, Brighid Blackman, Grace Dennis
References
1. Sullivan R, Alatise OI, Anderson BO, Audisio R, Autier P, Aggarwal A, et al. Global cancer surgery: delivering safe, affordable, and timely cancer surgery. Lancet Oncol. 2015;16(11):1193–1224.
2. Global Burden of Disease Cancer Collaboration, Fitzmaurice C, Allen C, Barber RM, Barregard L, Bhutta ZA, et al. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life-years for 32 Cancer Groups, 1990 to 2015: A Systematic Analysis for the Global Burden of Disease Study. JAMA Oncol. 2017 Apr 1;3(4):524.
3. Alkire BC, Raykar NP, Shrime MG, Weiser TG, Bickler SW, Rose JA, et al. Global access to surgical care: a modelling study. Lancet Glob Health. 2015 Jun 1;3(6):e316–23.
Publications:
Effects of hospital facilities on patient outcomes after cancer surgery: an international, prospective, observational study', The Lancet Global Health, 10 e1003-e1011 (2022)
SUNRRISE TRIAL 2019-2020
Single Use Negative pRessure dressing for Reduction In Surgical site infection following Emergency laparotomy
Australia and New Zealand Clinical Trials Registry Trial ID: ACTRN12619000496112
2019 MRFF International Clinical Trial Collaborations Grant $783,653
STORCC Team Members for SUNRRISE Study:
Andrew Drane, Elizabeth Lun, Sean Park, Kenneth Chew, Sara Clark, Winy Widjaja, Eu Jhin Lo, A Dawson
Publications:
CovidSurg 2020
CovidSurg was a platform of studies aiming to explore the impact of COVID-19 in surgical patients and services. Our studies were designed and delivered by an international collaborating group of surgeons and anaesthetists which reached more than 80 countries. CovidSurg – Globalsurg
Five cohort studies and one major survey have been run by CovidSurg:
• CovidSurg Cohort study: Included patients undergoing any type of surgery who had
SARS-CoV-2 infection in the 7 days before or the 30 days after surgery. Patients were
included from the start of the pandemic to 31 July 2020.
• CovidSurg-Cancer study: Included adult patients who required cancer surgery.
Patients were included from the start of the pandemic, up to 31 August 2020 at the
latest.
• GlobalSurg-CovidSurg Week study: Included patients undergoing any type of
surgery. Patients were included from 1 October 2020 to 31 October 2020.
• CovidSurg-3: Patient-level component: Included patients undergoing any type of
surgery who had SARS-CoV-2 infection in the 7 days before or the 30 days after
surgery. Patients were eligible for inclusion if they were operated 13 December 2021
to 28 February 2022.
• CovidSurg-3: Hospital-level component: Captured data on perioperative SARS-
CoV-2 rates and aggregated case-mix data at a hospital level. Patients were eligible
for inclusion if they were operated 1 December 2021 to 28 February 2022.
• Global surgery resilience index: Survey open for completion between 21 June to 1
August which measured the strength of elective surgery systems in your hospital.
CovidSurg: a cohort study, aiming to assess the outcomes of surgery in patients diagnosed with COVID-19
STORCC Team Members for CovidSurg Study:
A Dawson, Andrew Drane, Elizabeth Lun
Publications:
Early outcomes and complications following cardiac surgery in patients testing positive for coronavirus disease 2019: An international cohort study J Thorac Cardiovasc Surg 33933259
Machine learning risk prediction of mortality for patients undergoing surgery with perioperative SARS-CoV-2: the COVIDSurg Mortality Score British Journal of Surgery 34227657
Outcomes after peri-operative SARS-CoV-2 infection in patients with proximal femoral fractures: an international cohort study BMJ Open 34848515
Impact of COVID-19 on vascular patients worldwide: analysis of the COVIDSurg data Journal of Cardiovascular Surgery The Journal of Cardiovascular Surgery 2021 December;62(6):558-70 DOI: 10.23736/S0021-9509.21.12024-5
Mortality and pulmonary complications in emergency general surgery patients with COVID-19: a large international multicenter study Journal of Trauma and Acute Care Surgery: July 2022 - Volume 93 - Issue 1 - p 59-65
doi: 10.1097/TA.0000000000003577
Impact of Bacillus Calmette-Guérin (BCG) vaccination on postoperative mortality in patients with perioperative SARS-CoV-2 infection BJS Open 35022673
CovidSurg-Cancer: a cohort study assessing the safety of surgery for all types of cancer during the COVID-19 pandemic and the impact of the pandemic in cancer delay and treatment pathways.
STORCC Team Members for CovidSurg Cancer Study:
A Dawson, Andrew Drane, (Gosford Hospital)
A Dawson, Andrew Drane, Elizabeth Lun (Wyong Hospital)
Publications:
Effect of COVID-19 pandemic lockdowns on planned cancer surgery for 15 tumour types in 61 countries: an international, prospective, cohort study Lancet Oncology 34624250
Death following pulmonary complications of surgery before and during the SARS-CoV-2 pandemic British Journal of Surgery PubMed ID pending British Journal of Surgery, Volume 108, Issue 12, December 2021, Pages 1448–1464, https://doi.org/10.1093/bjs/znab336
The impact of surgical delay on resectability of colorectal cancer: an international prospective cohort study Colorectal Disease 35286766
Outcomes of gynecologic cancer surgery during the COVID-19 pandemic: an international, multicenter, prospective CovidSurg-Gynecologic Oncology Cancer study American Journal of Obstetrics and Gynecology 35779589
GlobalSurg- CovidSurg Week: a global cohort study aiming to determine the optimal timing to undergo surgery after a SARS-CoV-2 infection.
GlobalSurg CovidSurg network
122 Countries
1677 Centres
142,815 Patients
STORCC Team Members for CovidSurg Week Study:
Consultants: S Laura, S Clark, S Bengeri, P Stewart, B Munro, P Hamer, A Tchen, K Kwok, E Latif, K Wong, P Chen, R Poon, Z Hou, I Gunawardena, R McGee, D Wong, B Short, LH Le, A Dawson,
Andrew Drane, Elizabeth Lun, K Tree, U Pahalawatta, J Ma, H Narroway, T Ewington, K Chew, C Zhang, A DeBiasio, I Liang, V Lee, TY Ngan, J Kane, S Khanijaun, H Luo, P Ghosal, D Steiner, E Taylor, A Chong, B Buckland, S Van Ruyven, M Kaufman, C Parkin, H Cheah, S Miles, S Somasundran, W Ziaziaris, B Julien, D Jolly, B Mortimer, A Noor, T Cordingley, M Zhang, YS Lee, D Abulafia, EJ Loh, K Tran
Medical Students: E Devan, L Buith-Snoad, R Kaul, S Thong, V Yu, C Leung, C Saab, P Lin, M Park, S Fitt, V Ly, B Zhu, S Sebastion, R Simpson, S Holmes, T Khandelwal, R Amoils, N Bahtigur, A Gojnich, B Macnab, A Fatima, A Middleton, L Vance, J Gaul, P Ireland
Publications:
Timing of surgery following SARS-CoV-2 infection: an international prospective cohort study. Anaesthesia 33690889
SARS-CoV-2 vaccination modelling for safe surgery to save lives: data from an international prospective cohort study British Journal of Surgery 33761533
Effects of preoperative isolation on postoperative pulmonary complications after elective surgery Anaesthesia 34371522
SARS-CoV-2 infection and venous thromboembolism after surgery: an international prospective cohort study Anaesthesia 34428858
Prize:
“Timing of surgery following SARS-CoV-2 infection: an international prospective
cohort study” awarded Paper of the Year 2021 by Anaesthesia.**Highest-ever Altmetric score for this journal, as of July 2021.
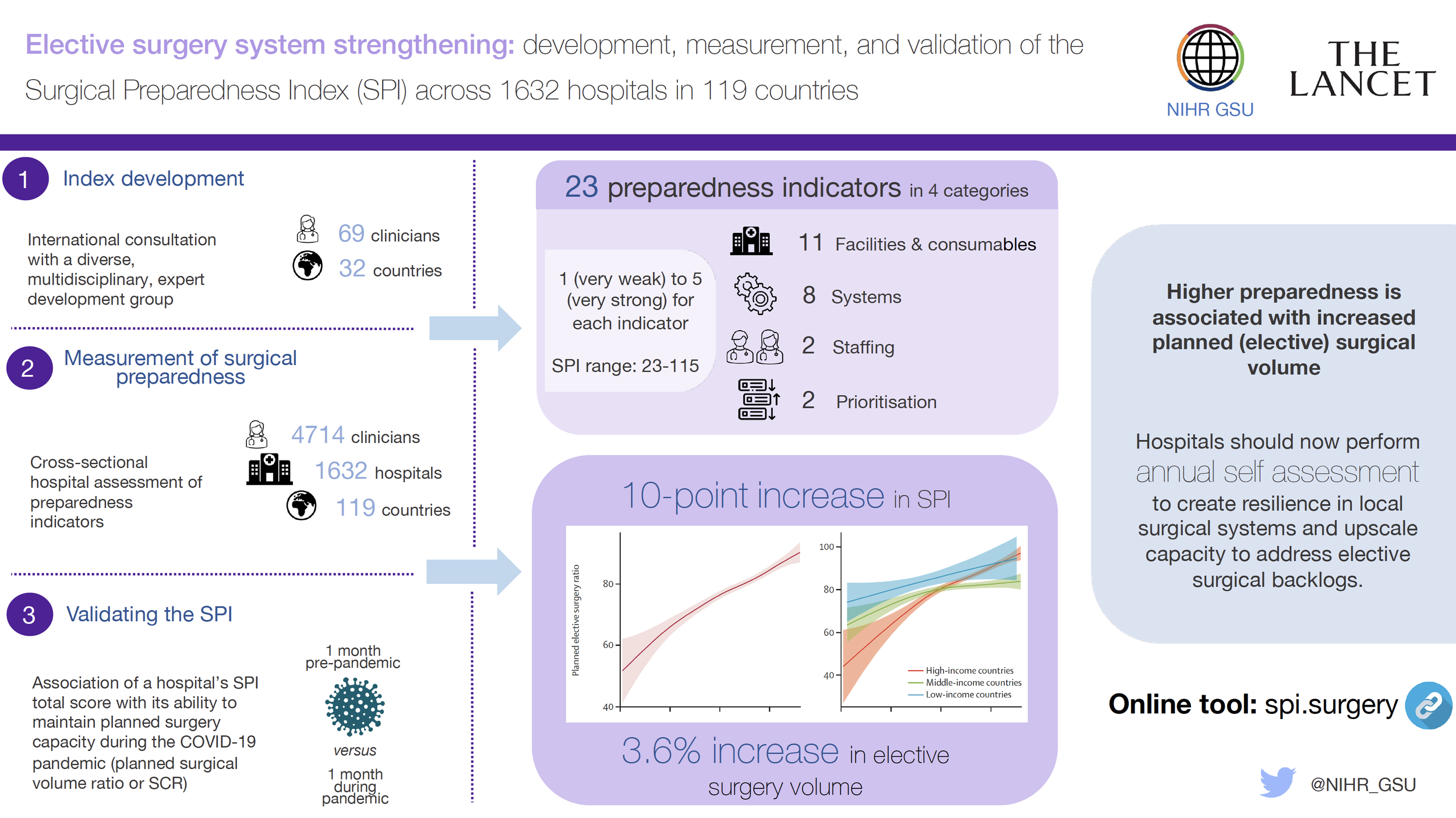
GlobalSurg and Covidsurg System Preparedness Index
Cancellations of planned (elective) surgery were common during COVID-19, with a global elective surgery crisis now facing health systems worldwide. However, there are many examples of pressures on health systems from weather events, to seasonal pressures, natural disasters and warfare.
The System Preparedness Index (SPI) is a simple tool that can help you prevent cancellations of surgery in the future by making your local service better prepared for times when it is under pressure. it has been validated by 4,714 clinicians in 1,632 hospitals across 119 countries.
Key finding: Surgical systems worldwide were poorly prepared for external stresses such as COVID-19.
A 10-point increase in your hospital's SPI score could help 4 more patients have surgery per 100 patients on the waiting list.
Collaborators: Amanda C Dawson, Sharon, E, Laura, Elizabeth W Y Lun, Ina X Liang (Gosford Hospital) Kevin Tree (Wyong Public Hospital).
Publications:
Elective surgery system strengthening: development, measurement, and validation of the Surgical Preparedness Index (SPI) across 1632 hospitals in 119 countries The Lancet 31 October 2022 DOI:https://doi.org/10.1016/S0140-6736(22)01846-3 https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(22)01846-3/fulltext
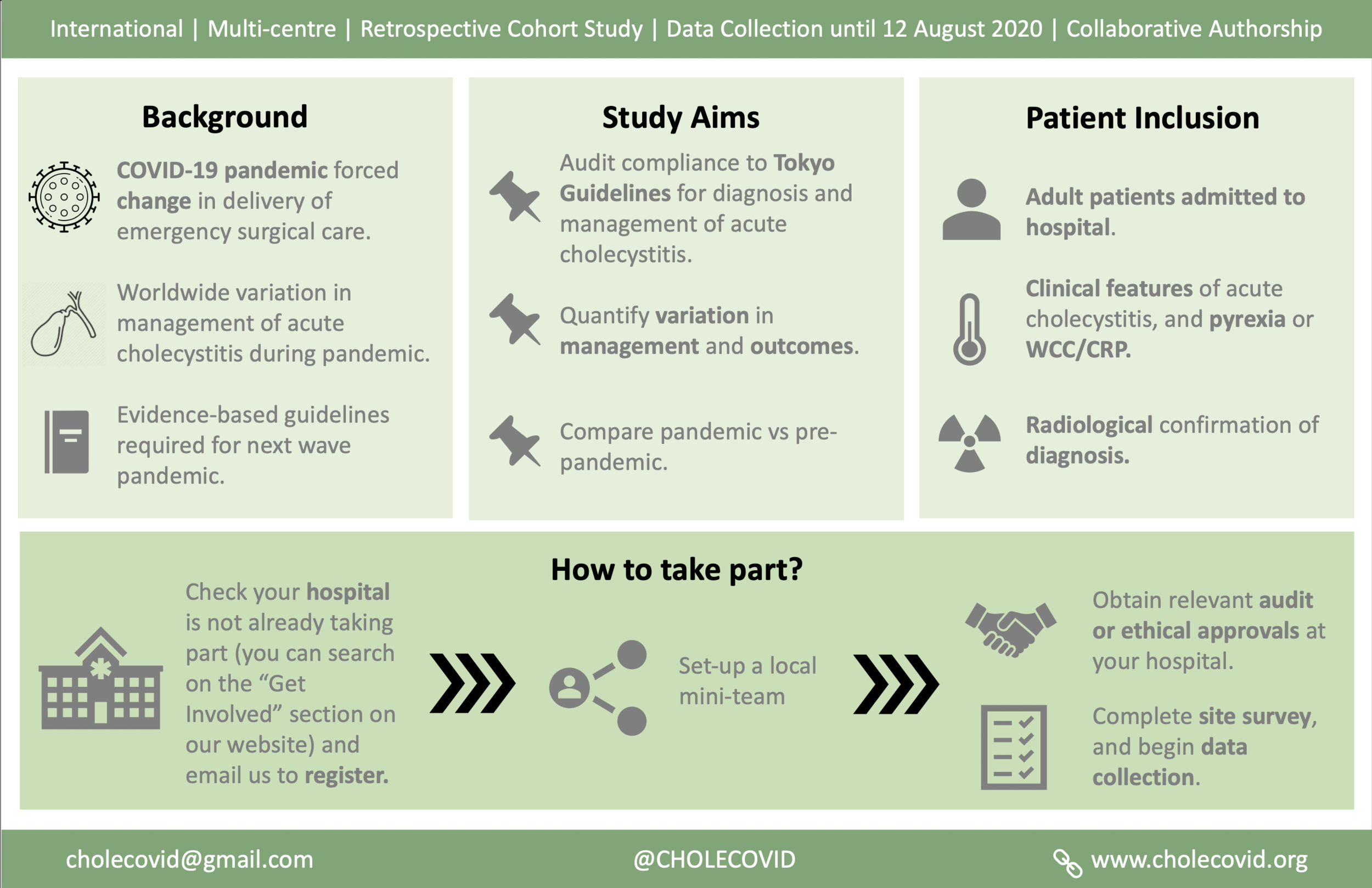
Cholecovid 2020
International multi-centre appraisal of the management of acute CHOLEcystitis during the COVID-19 pandemic
STORCC Team Members for CHOLECovid Study:
A Dawson, Andrew Drane, Elizabeth Lun, Stephanie Van Ruyven
Publications:
Global overview of the management of acute cholecystitis during the COVID-19 pandemic (CHOLECOVID study) CHOLECOVID Collaborative BJS OPEN 2022 May 2;6(3):zrac052. doi: 10.1093/bjsopen/zrac052.
PROJECT STATUS: DATA ANALYSIS:
COVIDCare Collaborative Project
STORCC was pleased to participate in the COVIDCare Collaborative Project. This was a nationwide survey of all surgical trainees to understand the effects of the COVID-19 pandemic on their training, wellbeing and home life run by the VERITAS Collaborative. VERITAS (Victorian collaborative for Education, Research, Innovation, Training and Audit by Surgical trainees) was established at University of Melbourne, Department of Surgery – Austin Precinct, in 2018,
POSTVenTT Trial
Postoperative variations in anaemia treatment and transfusions (POSTVenTT), a prospective multicentre observational cohort study of anaemia after major abdominal surgery
This is a clinical audit, which will involve mini-teams of 1 – 4 collaborators per speciality group per data collection period. These teams will collect data over two continuous 14-day periods on all consecutive patients undergoing major surgery at participating hospitals, with a follow-up at 30 postoperative days. A consultant will supervise all mini-teams.
Primary Aim:
To audit compliance with pre-, intra-, and postoperative guidelines for management of anaemia in patients undergoing major abdominal surgery.
Secondary Aims:
To characterise incidence of anaemia following major abdominal surgery.
To identify risk factors associated with postoperative anaemia.
To explore associations between postoperative anaemia and rate of postoperative complications.
To explore association of postoperative anaemia with short-term outcomes (length of stay, readmission to Hospital).
To audit the use or iron therapy or blood transfusion.
STORCC Team for POSTvenTT:
Regional leads Australia:
Amanda C Dawson, Elizabeth WY Lun (New South Wales);
Supervising consultants Australia:
Amanda C Dawson (Gosford Hospital); Richard G McGee (Wyong Hospital)
Hospital Leads:
Amanda C Dawson, Elizabeth WY Lun (Gosford Hospital); Richard G McGee (Wyong Hospital).
Data collectors:
Sneha E Ninan, Gemma J Qian, Joseph W Fletcher, Brendan Watkins, Luke C Green, Yara A Elgindy, Elodie V Honore, Charles J Roth, Jonathan Tandjung, Adam R Perkovic, Luca Borruso, Victor Yu, Thomas-Hoang Le, Samuel M Mathias, Daniel Jeong, Charlotte E Cornwell, Emily A Taylor, Kie Ren Oon (Gosford Hospital)
Rosalina Lin, Jade T Bahnisch, Haili Luo (Wyong Hospital)
Publications:
The management of peri-operative anaemia in patients undergoing major abdominal surgery in Australia and New Zealand: a prospective cohort study. Med J Aust. 2022 Nov 7;217(9):487-493. doi: 10.5694/mja2.51725. Epub 2022 Sep 22. PMID: 36134647.
Opioid prescription patterns and usage across Australia and Aotearoa New Zealand: A prospective, multi-centre cohort study (oPERAS)
OPERAS is an international, prospective multi-centre cohort study that aims:
To compare the type, quantity and duration of postoperative, post-discharge opioid prescriptions for postoperative analgesia to what is consumed by patients at 7-days post-discharge
To describe the variations in opioid prescription and consumption after discharge from surgery
To identify risk factors for opioid overprescribing after common surgical procedures
Through the OPERAS Study, we aim to build collaborative research capacity within medical students and junior doctors in ANZ.
STORCC Team for OPERAS:
OPERAS Scientific Advisory Group: Amanda Dawson, Associate Professor, Surgery (University of Newcastle, Newcastle, Australia);
Publications:
Opioid PrEscRiptions and usage After Surgery (OPERAS): protocol for a prospective multicentre observational cohort study of opioid use after surgery
BMJ Open 2022;12:e063577. doi: 10.1136/bmjopen-2022-063577
Funding OPERAS has received funding from the Maurice and Phyllis Paykel Trust (Ref: 213122) and Surgical Research Funds, University of Newcastle.
PROJECT STATUS: DATA ANALYSIS:
SNAP-3 to examine
Frailty, delirium and the older surgical patient
The 3rd Sprint National Anaesthesia Project (SNAP-3) is an observational cohort study that aims to describe the impact of frailty and delirium, and their management, on outcomes following surgery in older people. In common with SNAP-1 and SNAP-2 it will consist of a short period of data collection from potentially every eligible patient having surgery in a defined period of time. We estimate that approximately 12,000 individuals will take part, making it one of the largest studies of its kind in the world.
We hope the results of the study will help improve decision making by patients and clinicians, as well as providing evidence for optimal design of perioperative services.
These pages will be updated regularly with progress and news. SNAP-3 is generously supported by the Royal College of Anaesthetists. It is being jointly managed by the Health Services Research Centre and the University of Nottingham.
COVIDCare
Collaborative Project
STORCC was pleased to participate in the COVIDCare Collaborative Project. This was a nationwide survey of all surgical trainees to understand the effects of the COVID-19 pandemic on their training, wellbeing and home life run by the VERITAS Collaborative. VERITAS (Victorian collaborative for Education, Research, Innovation, Training and Audit by Surgical trainees) was established at University of Melbourne, Department of Surgery – Austin Precinct, in 2018. VERITAS (unimelb.edu.au)